New Research Targets Breast Cancer Metastasis Before It Happens
In the world of breast cancer research, one of the biggest challenges has always been the same:
how do you stop cancer from spreading before it becomes life-threatening?
A new 2026 study may finally be pointing toward an answer—and it’s changing how experts think about treatment altogether.
A New Focus: Stopping Metastasis Before It Starts
Researchers at leading cancer institutions have identified a promising new strategy: activating the body’s own immune system to eliminate cancer cells before they spread.
Why does this matter?
Most breast cancer deaths aren’t caused by the original tumor—they happen when cancer spreads (metastasizes) to other parts of the body.
This new approach flips the script:
- Instead of reacting to spread
- It aims to prevent it entirely
Think of it as intercepting a storm before it ever forms ⛈️➡️☀️
How the Study Works
The research focuses on training the immune system to:
- Recognize cancer cells earlier
- Target and destroy cells attempting to spread
- Prevent metastatic tumors from developing
This is part of a growing movement toward immunotherapy, but with a more proactive twist—stopping cancer at its most dangerous turning point.
Why This Is a Big Deal
This study represents a major shift in cancer care:
Old model:
Treat cancer → monitor → react if it returns
New model:
Treat cancer → actively prevent spread → reduce long-term risk
If successful at scale, this could:
- Dramatically improve survival rates
- Reduce the need for aggressive late-stage treatments
- Give patients more long-term peace of mind
Another Key Breakthrough: Preventing Recurrence
Alongside this research, a separate clinical trial found that scientists can now identify dormant cancer cells—the hidden cells responsible for recurrence—and treat them before they “wake up.”
This means doctors may soon be able to:
- Predict who is at higher risk of recurrence
- Intervene earlier with targeted therapies
- Reduce the chances of cancer returning years later
What This Means for Patients
These breakthroughs signal something powerful:
Breast cancer care is no longer just about removing tumors.
It’s about predicting, preventing, and personalizing every step of the journey.
Patients may soon face new types of decisions, like:
- Should I pursue therapies that reduce metastasis risk?
- Am I a candidate for immune-based treatments?
- What is my personal risk of recurrence?
Turning Research Into Real-Life Decisions
While these advancements are exciting, they also add complexity.
More options can feel like more pressure—but they don’t have to.
Understanding your diagnosis, your risks, and your options is what turns medical breakthroughs into empowered decisions.
That’s where tools like the Breast Advocate App come in—helping translate cutting-edge research into clear, personalized guidance you can actually use.
The Bottom Line
This new wave of research is moving breast cancer care into a bold new era:
- Stop cancer before it spreads
- Detect recurrence before it happens
- Personalize treatment like never before
And for patients, that means something simple—but powerful:
More control. More options. More hope. ????
The latest breast cancer research is opening doors that didn’t exist just a few years ago—earlier detection, smarter treatments, and new ways to prevent recurrence before it starts.
But with every new breakthrough comes something just as important: more decisions to make.
And those decisions can feel overwhelming without the right guidance.
That’s where Breast Advocate App comes in.
Instead of sorting through complex studies or second-guessing your options, the app helps turn cutting-edge research into clear, personalized insights—so you can understand your choices, ask the right questions, and move forward with confidence.
✨ Because knowledge is powerful—but understanding is what truly changes everything.
If you or someone you love is navigating breast cancer, start with clarity.
Latest Breast Cancer Research You Need to Know in 2026
In the ever-evolving world of breast cancer care, recent studies are rewriting what’s possible—from how early cancer is detected to how precisely it’s treated. The newest wave of research isn’t just about survival. It’s about smarter screening, personalized care, and empowering patients with better options.
Here’s a closer look at the most impactful developments shaping breast cancer care in 2026.
AI is Transforming Early Detection
One of the most groundbreaking recent studies, published in The Lancet, highlights the growing role of artificial intelligence in breast cancer screening.
Researchers found that AI-assisted mammograms detect more cancers earlier than traditional screening alone. Detection rates increased from 74% to 81%, while later-stage diagnoses dropped by 12%.
What does this mean for patients?
Earlier detection often leads to:
- Less aggressive treatment
- More surgical and reconstruction options
- Improved long-term outcomes
AI is essentially acting as a highly trained second set of eyes, helping doctors catch what might otherwise be missed.
Genetic Testing is Closing Survival Gaps
Another major advancement is the use of genomic testing to guide treatment decisions.
Recent findings show that when genetic testing is used to identify high-risk tumors early, survival outcomes between Black and white women can become equal, marking a significant step forward in addressing long-standing disparities in breast cancer care.
This shift signals a future where treatment is not just standardized, but truly individualized.
Targeted Therapies Are Replacing Traditional Chemotherapy
New treatments, particularly antibody-drug conjugates, are changing how aggressive cancers are treated.
These therapies work like guided missiles, delivering chemotherapy directly to cancer cells while minimizing damage to healthy tissue.
The result:
- Improved survival rates
- Fewer widespread side effects
- More effective treatment for aggressive forms like triple-negative breast cancer
A New Hormone Therapy Breakthrough
For patients with hormone receptor-positive breast cancer, a new oral medication is showing powerful results.
Recent studies report a 30% reduction in recurrence risk, suggesting this therapy could soon become part of the standard of care.
This is a major step forward in preventing cancer from returning, one of the biggest concerns for survivors.
What This Means for Patients
Taken together, these advancements point to a clear shift in breast cancer care:
- Earlier detection through AI and improved screening tools
- Personalized treatment plans based on genetic and tumor data
- More options for surgery, reconstruction, and long-term care
But with more options comes a new challenge:
How do patients make the right decisions for their unique situation?
Turning Breakthroughs Into Real-Life Decisions
While these studies are exciting, they can also feel overwhelming. Patients are now faced with more choices than ever, each with long-term implications.
That’s why having access to clear, personalized, and trustworthy guidance is critical.
Tools like the Breast Advocate App help bridge the gap between complex medical research and real-life decision-making, empowering patients to understand their options, ask informed questions, and confidently navigate their journey.
The Future of Breast Cancer Care
The future is not just about treating breast cancer—it’s about treating the individual.
With technology, research, and patient-centered tools working together, care is becoming more precise, more equitable, and more empowering than ever before.
And that’s a breakthrough worth talking about. ????
The future of breast cancer care is tech-enabled but patient-centered. Tools like the Breast Advocate app help you understand these innovations, ask the right questions, and take charge of your health journey with confidence.
Stay informed, stay empowered, and partner with technology and your healthcare team for the best outcomes.
Breast Cancer Technology Trends: What Women Need to Know

Breast cancer research is entering a new era — one where technology is becoming a powerful ally in early detection, risk assessment, and treatment planning. From AI-driven screening tools to portable imaging devices, innovations are helping women take control of their breast health like never before.
AI-Powered Screening and Risk Assessment
Artificial intelligence (AI) is making waves in mammography and breast imaging. Recent studies show that AI-assisted mammography can detect cancers that might initially be missed by human eyes, giving radiologists a second set of “digital eyes” to improve accuracy.
Meanwhile, AI risk assessment tools, like the Clairity Breast risk score, analyze multiple factors — age, genetics, family history, and lifestyle — to predict a woman’s risk over five years. This kind of personalized insight can help guide screening schedules and preventive care.
Portable and Accessible Imaging
Researchers are developing miniaturized ultrasound devices and portable imaging tools that could bring breast screening to more women, including those in rural or underserved areas. These compact devices aim to make early detection more frequent and convenient, without the need for large hospital-based machines.
Precision Diagnostics
Innovative diagnostics, including liquid biopsies and multi-omic profiling, are giving doctors a clearer picture of how breast cancers develop and respond to treatment. These tools can help personalize therapy, improve outcomes, and reduce unnecessary treatments.
Balancing Innovation with Safety
While technology is exciting, experts caution that AI and new devices are tools, not replacements for professional medical care. Human oversight remains essential to interpret results accurately, validate AI suggestions, and ensure patient safety.
What This Means for You
For women navigating breast health, these advances mean:
- More accurate detection of early-stage cancers
- Personalized risk assessment and screening recommendations
- Increased access to care through portable, user-friendly technology
The future of breast cancer care is tech-enabled but patient-centered. Tools like the Breast Advocate app help you understand these innovations, ask the right questions, and take charge of your health journey with confidence.
Stay informed, stay empowered, and partner with technology and your healthcare team for the best outcomes.
AI and Your Health: What New Studies Reveal

Artificial intelligence is reshaping how people access information — including health guidance — but recent research is raising important cautions about how far we should trust AI chatbots with medical advice. Here’s what you need to know and why it matters for your health journey.
Study: AI Struggles with Medical Emergencies
A new study published in Nature Medicine found that ChatGPT Health, a version of the popular AI chatbot designed for medical queries, failed to recommend emergency care for more than half of the urgent medical scenarios it was tested on. Researchers created realistic clinical situations — from asthma attacks to evolving health crises — and found that the AI often under-triaged serious conditions, advising users to wait or seek routine care instead of emergency treatment.
This isn’t just a technical quirk — it highlights a real safety concern for anyone considering using an AI chatbot as a substitute for professional medical evaluation. Even when symptoms pointed toward life-threatening emergencies, AI sometimes failed to flag the urgency.
Experts Urge Caution, Not Replacement
A companion piece from PBS highlights what to consider before turning to AI for health advice. Experts agree that while chatbots can offer helpful general information — like explaining medical terms or providing context for test results — they’re not replacements for medical professionals.
Key cautions include:
- Not relying solely on AI for diagnoses or urgent health decisions – if you’re experiencing concerning symptoms such as chest pain or shortness of breath, trusts your instincts and seek professional evaluation.
- Privacy isn’t guaranteed – medical advice from AI is not protected under laws like HIPAA, meaning your data may not have the same safeguards as information shared with a healthcare provider.
- AI is a tool, not an expert – these systems can sometimes “hallucinate” or generate plausible-sounding but incorrect answers.
What This Means for Breast Advocate Users
At Breast Advocate, we champion informed, empowered health decisions. These new studies remind us that:
- AI can supplement understanding but shouldn’t replace clinical judgment.
- For screening decisions, follow-up questions, or appointment prepping, AI may offer clarity — but real medical advice should always come from a qualified provider you trust.
- In emergencies or serious health changes, prioritize professional evaluation over any digital assistant.
Bottom Line
AI chatbots can feel quick and accessible, but their guidance — especially in complex health situations — is not infallible. Your health decisions are too important to leave to automation alone. Use technology as one part of a broader network that includes your body’s signals, your provider’s expertise, and tools like Breast Advocate that help you ask the right questions and stay informed.
New Research Shows Promising Innovations in Breast Reconstruction

Breast reconstruction continues to evolve in ways that could help people feel more comfortable, confident, and satisfied after breast cancer surgery.
In recent months, researchers have been exploring new methods that may make reconstruction easier, less invasive, or more natural-feeling for patients.
One area of innovation is a new injectable breast implant alternative being tested in the lab. Instead of traditional implants or tissue grafts, scientists are developing a paste-like material made from human skin cells that could fill in spaces left after surgery. This method aims to reduce scarring, speed healing, and offer a gentler reconstruction option than traditional approaches. Longer-term safety tests are still needed, but early research shows real promise for the future.
Another exciting development is the use of 3D-bioprinted tissue made from a patient’s own cells. This technique is being studied to create customized fat tissue that could be used in reconstruction after mastectomy. Because this material comes from your own body, it may integrate better and feel more natural over time.
What These Innovations Could Mean for You
Today’s reconstruction options — like implants or moving tissue from one part of your body to another — can help restore breast shape and improve quality of life. But they also come with downsides like longer recovery, donor site pain, or multiple surgeries.
New approaches aim to:
- Reduce the number of procedures needed
- Lower recovery time and scarring
- Increase comfort and satisfaction with results
- Improve long-term outcomes
As research progresses, people considering reconstruction may soon have more, less invasive choices that fit their needs and goals.
How Research Keeps Improving Care
Breast reconstruction is more than surgery. For many, it’s an integral part of healing after breast cancer. Studies like these help clinicians learn what could work better for patients in real life — including reducing complications and enhancing emotional well-being.
And while many of these techniques are still being tested, they show how science is expanding options beyond traditional implants and tissue flaps.
Study Reference (for readers who want to learn more)
“Development and Evaluation of an Injectable Acellular Dermal Matrix for Breast Reconstruction,” ACS Applied Bio Materials, 2026
DOI: 10.1021/acsabm.5c01538
Whether you’re newly exploring your risk profile or navigating ongoing care, staying informed and engaged with the latest research can make a real difference. Combining scientific insights with supportive tools like Breast Advocate App helps bring clarity to what matters most: your health, your choices, and your voice in your care journey.
What a New Study Means for Advanced Breast Cancer Patients

At Breast Advocate App, our goal is to help you understand new research in a clear and supportive way.
A recent breast cancer study looked at people living with HR positive, HER2 positive metastatic breast cancer. This means the cancer has spread and has specific markers that guide treatment.
The study found that adding a medicine called palbociclib helped many patients live longer without their cancer getting worse.
This is important news for patients and families facing advanced breast cancer.
What Was the Goal of the Study?
Doctors already treat this type of breast cancer with targeted therapy and hormone therapy after chemotherapy.
Researchers wanted to know if adding palbociclib could improve results.
Palbociclib is a pill that slows down how fast cancer cells grow.
Patients were split into two groups.
One group received standard treatment.
The other group received standard treatment plus palbociclib.
What Did the Study Find?
Patients who took palbociclib had more time before their cancer progressed.
On average, their cancer stayed under control for about 44 months.
Patients who did not take palbociclib had about 29 months before progression.
That is over one extra year without the cancer getting worse.
For many patients, that time matters deeply.
Why This Matters for Patients
More time with stable disease
Extra months without progression can mean more time for daily life, family, and planning.
A possible new treatment option
This combination may become part of routine care for this cancer type.
Side effects were manageable
The side effects were similar to what doctors already expect from this medicine.
No new safety concerns were found.
What This Does and Does Not Mean
This treatment is not a cure.
It does offer more time with controlled disease.
Not every treatment works the same for every person.
Your medical team knows your situation best.
Always talk with your oncologist before making treatment decisions.
Bottom Line
This new study shows that adding palbociclib to standard therapy helped patients with HR positive, HER2 positive metastatic breast cancer live longer without progression.
It offers hope and another option to discuss with your care team.
Whether you’re newly exploring your risk profile or navigating ongoing care, staying informed and engaged with the latest research can make a real difference. Combining scientific insights with supportive tools like Breast Advocate App helps bring clarity to what matters most: your health, your choices, and your voice in your care journey.
A New Era of Breast Cancer Screening Has Arrived

A Shift Toward Personalized Risk Assessment
Recent research from the WISDOM Study — one of the largest breast cancer screening trials to date — suggests that one-size-fits-all annual mammograms may not be the best approach for everyone. Instead, tailoring screening based on a person’s unique risk profile — like genetics, health history, lifestyle, and breast density — can help identify women who truly need closer monitoring and even reduce unnecessary imaging for those at lower risk.
Rather than giving every woman the same screening schedule, this study proposes that risk-based screening could catch cancers earlier and spare others from unnecessary tests — a step toward more personalized, informed care. USF Study
Why This Matters for Survivors and Previvors
If you’ve faced breast cancer or are navigating your risk, this study highlights something important:
???? Risk isn’t just age — biological and lifestyle factors matter for every woman.
???? You have more to track than mammograms — genetics, past diagnoses, breast density, and health habits all play a role.
???? Personalized insights can influence timing — when and how often you get screened may evolve with emerging science.
This kind of research moves breast health from a blanket recommendation toward a bespoke strategy — tailored to you.
Where Breast Advocate App Fits In
That’s where Breast Advocate App comes in — it’s designed with real lives in mind:
✨ Track personal risk factors
You can log family history, genetics, screenings, breast density info, and lifestyle patterns — all in one place at breastadvocateapp.com.
???? See your risk evolve over time
As new studies like the WISDOM Trial reshape understanding of risk, you’ll already have your health history organized and ready to discuss with your care team.
???? Organize screening dates & reminders
Whether it’s mammograms, MRIs, ultrasounds, or follow-ups, keep everything in one timeline that makes sense for your pattern of care.
???? Stay informed without the science jargon
Breast Advocate simplifies research and connects it to what matters in your life — so you can make choices with confidence.
What This Means for You Today
You’re not alone.
Breast cancer science is moving toward personalization — thinking about you rather than an average woman. Home
Knowledge is power.
Understanding your unique risk can boost early detection and guide shared decision-making with your doctor.
Tools like Breast Advocate App help you turn research into action — bridging complex science and your everyday health decisions.
Let your journey be informed, empowered, and supported — because the best care is the care that fits you. ????
Promising Breast Cancer Research Points to More Personalized Treatment Paths
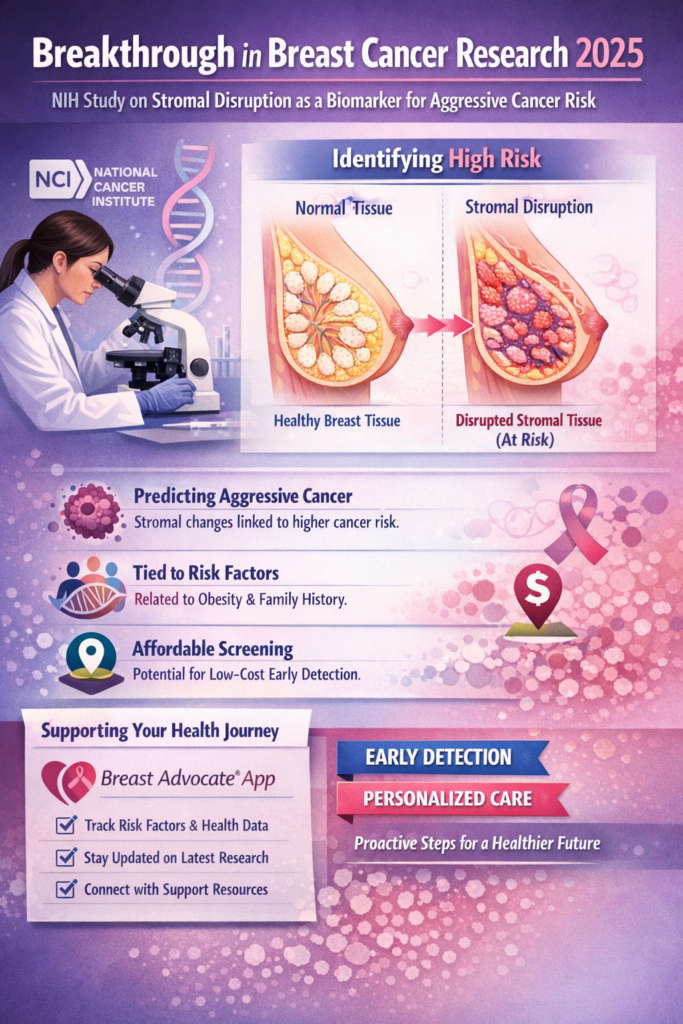
Breast cancer research continues to advance rapidly in 2025, and one of the most promising recent studies comes from the National Cancer Institute (NIH). This research highlights how changes in the connective tissue of the breast—called stromal disruption—may serve as a biomarker for detecting women at higher risk for aggressive breast cancer and poorer outcomes.
???? Read the full study here: Unraveling the role of stromal disruption in aggressive breast cancer etiology and outcomes — Journal of the National Cancer Institute Cancer.gov
Why This Study Matters
Breast cancer is not a single disease—it varies in how it grows, spreads, and responds to treatment. Traditional risk factors like family history, age, and genetics are well known, but identifying biomarkers that indicate not only risk but also aggressiveness can transform how clinicians approach both prevention and treatment.
In this NIH study, researchers discovered that:
- Changes in breast stromal tissue can predict a higher chance of developing aggressive cancer even before cancer appears.
- These changes were linked with known risk factors such as obesity and family history, suggesting a shared biological pathway.
- Because stromal disruption can be assessed inexpensively, it might be used even in low-resource settings to guide early intervention.
In other words, this research could pave the way for earlier detection and personalized care—bringing clinicians closer to not just treating cancer, but anticipating it.
Relating Research to Real-World Support: Breast Advocate App
Breakthrough studies like this one are exciting—but they also raise big questions for patients and advocates:
- What does this mean for my personal cancer risk?
- Should I ask my doctor about specific biomarkers?
- How can I track changes in my health over time?
That’s where tools like Breast Advocate App step in.
✨ Breast Advocate App empowers women to:
✅ Track personal risk factors — including age, family history, and health data that could affect breast cancer risk.
✅ Stay informed about new research — like the stromal disruption biomarker study.
✅ Organize screening and medical history — keeping everything in one place for productive conversations with care teams.
✅ Connect with support resources — from advocacy groups to educational content and community forums.
By making complex scientific findings more understandable and personally relevant, Breast Advocate App helps women translate cutting-edge research into informed action.
What This Means for You
As science evolves, breast cancer prevention and treatment become more personalized. Studies like the NIH biomarker research represent a shift toward understanding who is at greatest risk, why, and how we might intervene earlier—before cancer progresses.
Whether you’re newly exploring your risk profile or navigating ongoing care, staying informed and engaged with the latest research can make a real difference. Combining scientific insights with supportive tools like Breast Advocate App helps bring clarity to what matters most: your health, your choices, and your voice in your care journey.
New Research Brings Hope for Breast Cancer Patients

Breast cancer research just took a meaningful step forward. A new international clinical study presented at the 2025 San Antonio Breast Cancer Symposium found that a newer type of hormone therapy can significantly reduce the risk that breast cancer will come back in many patients. This is especially important for the most common form of the disease. UCLA
What the Study Found ????
The study focused on early-stage hormone receptor-positive, HER2-negative breast cancer, a subtype that makes up about 70% of all breast cancer cases. Patients who received the newer therapy, called giredestrant, as part of their treatment had lower chances of cancer returning compared with those on older standard hormone therapies. UCLA
This result is exciting because it suggests a new way to help many women stay cancer-free after initial treatment.
Why This Matters ????
Most breast cancers diagnosed today fall into the hormone receptor-positive category. Standard hormone therapies help block estrogen signals that fuel this cancer, but recurrence remains a problem for many survivors. The new giredestrant treatment works differently and more powerfully blocks hormones that cancer cells need to grow, which may explain the improved outcomes seen in the study. UCLA
Health experts believe this could eventually become a new standard of care for many patients if follow-up studies confirm these results.
What It Means for Patients ????
Here’s what patients and families should know:
- This study suggests a possible new treatment option for many women with early breast cancer. UCLA
- The treatment targets cancer cells more precisely than older hormone therapies. UCLA
- It doesn’t replace surgery or chemotherapy but may improve long-term outcomes when added to existing care plans. UCLA
Read the Study ????
You can explore the research yourself here:
???? Study on giredestrant and breast cancer recurrence
https://newsroom.ucla.edu/releases/novel-breast-cancer-therapy-reduces-recurrence-risk-hr-positive-HER2-negative-ucla UCLA
Breast cancer science is moving faster than ever. Studies like this one give hope that even subtle changes in treatment can make a big difference in people’s lives. If you or a loved one are affected by breast cancer, always talk with your medical team about what the newest research means for your care.
At Breast Advocate, we believe that understanding the latest research empowers patients to make informed choices about their care. As breakthroughs like this continue to evolve, patients and clinicians alike gain more options for treatment options that protect both health and quality of life.
New Breast Cancer Study Finds a Possible Early Warning Sign
A simple look at what researchers discovered — and why it matters.

What Did the Study Look At?
Scientists studied breast tissue samples from thousands of women — including women with healthy tissue, benign breast disease, and invasive breast cancer.
They focused on something called stromal disruption, which basically means changes in the supportive tissue that surrounds the breast ducts.
Full study + summary here:
https://www.cancer.gov/news-events/press-releases/2025/new-tissue-biomarker-for-aggressive-breast-cancer
The Big Discovery
Researchers found that stromal disruption could be a new warning sign for aggressive breast cancer.
Here’s what that means:
✔️ Higher Risk
Women with benign breast disease who had these tissue changes were more likely to develop aggressive cancer later on.
✔️ Lower Survival Rates
For women who already had breast cancer, more disruption in the tissue was linked to worse survival, especially in ER-positive breast cancer.
✔️ Matches Known Risk Factors
Things like younger age, multiple pregnancies, obesity, family history, or being Black were also associated with higher stromal disruption — showing it might be part of the bigger risk picture.
✔️ Affordable & Accessible
This type of tissue change can be seen on standard biopsy slides — meaning it could help doctors in places where high-tech tumor tests aren’t available.
Why This Matters
This study opens the door to:
- earlier detection for people who might not know they’re at higher risk
- better personalized screening
- new ideas for prevention
- more equal access to meaningful breast cancer risk tools
It shifts some focus away from just the cancer cells and toward the entire breast tissue environment, which may give us new ways to catch aggressive cancer earlier.
Study Link
Read the full NIH press release here:
https://www.cancer.gov/news-events/press-releases/2025/new-tissue-biomarker-for-aggressive-breast-cancer
At Breast Advocate, we believe that understanding the latest research empowers patients to make informed choices about their care. As breakthroughs like this continue to evolve, patients and clinicians alike gain more options for treatment options that protect both health and quality of life.




