5 Things You Should Know Before Having Breast Reconstruction Surgery

Breast implants are NOT the only option
The most commonly performed method of breast reconstruction performed today uses tissue expanders and implants. Although this approach is a good option for many, it’s not the only option. Likewise, implants may not be the best option for some patients. Reconstruction options using your own tissue (referred to as autologous or “flap” reconstruction) is also an option. In particular, after radiation treatment flap procedures are associated with fewer complications than implant-based reconstructions.
Sensory nerve reconstruction may be an option
Following a mastectomy, many patients experience permanent numbness to the chest area and reconstructed breast. This is because the sensory nerves that provide feeling are usually cut during the mastectomy. There is some good news though! Advances in breast reconstruction techniques have made sensory nerve reconstruction possible: reconnecting the sensory nerves in the chest can significantly improve the return of feeling to the reconstructed breast.
Enhanced Recovery After Surgery (ERAS) protocols are making recovery easier
Many surgeons are now implementing ERAS protocols to ensure their patients experience an easier recovery following breast cancer surgery, with or without reconstruction. Regardless of the type of reconstruction performed, ERAS protocols are reducing hospital stays, shortening recovery, and reducing the need for narcotics to control discomfort after surgery.
Shared decision-making matters
Breast reconstruction is not a one-size-fits-all procedure. Ensuring you discuss all your options and how they align with your lifestyle, preferences, and goals is critical in planning the best reconstructive option for you.
You can choose NOT to have breast reconstruction
It is important for patients to remember choosing NOT to undergo breast reconstruction and instead opting to “go flat” is an acceptable choice. Going flat (aesthetic flat closure) gives patients seeking no reconstruction the ability to maintain balance and symmetry without reconstructing the breast(s). Women can also choose to go flat after breast reconstruction if they are unhappy with their reconstruction results or have experienced complications after breast reconstruction.
To learn more about ALL your options, download the Breast Advocate App today!
BETting Against Brain Mets With Promising Combination Therapy
Breast cancer metastasis (also known as “stage IV”) occurs when cancer cells leave the breast and travel to other parts of the body in the bloodstream or via the lymphatic system. The most common sites of spread are the liver, brain, bones, and lungs. About 30% of women diagnosed with early-stage breast cancer will ultimately develop metastatic disease. About 55% of women with HER2-positive breast cancer will progress to stage 4. Metastasis greatly impacts long-term survival; for breast cancer patients whose cancer has metastasized to the brain, the life expectancy is only about six months.
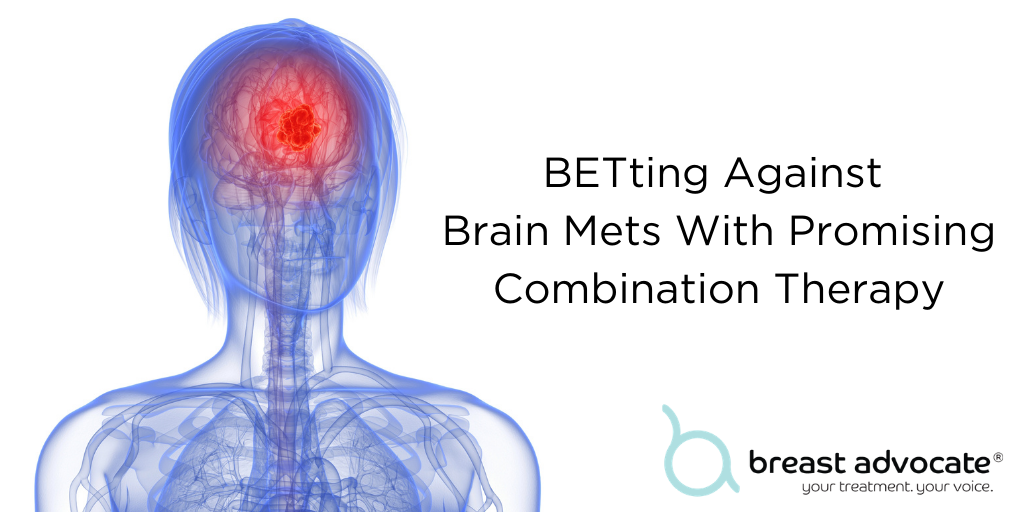
Brain metastases are difficult to reach and often are not susceptible to the same treatments as breast cancers in other parts of the body because they are blocked by the blood-brain barrier. However, a new study from Northwestern Medicine is reports some positive findings on a new treatment option.
A new combination therapy including a class of drug known as a BET inhibitor, greatly decreased the size of brain mets and increased survival in mice. About 75% of the mice who underwent this new treatment were cancer-free following the treatment. The BET inhibitor appears to sensitize breast cancer brain metastases to vinorelbine, a drug already approved by the FDA, demonstrating a potentially very promising therapeutic combination.
“The new combination therapy we identified can cross the blood-brain barrier,” said lead study author Dr. Maciej Lesniak, Northwestern Medicine chair of neurological surgery and professor of neurosurgery at Northwestern University Feinberg School of Medicine. “The therapy also targets brain metastases and significantly improves survival.”
The drug I-BET-762, used in combination with vinorelbine in the study, is only approved for trials by the FDA at this time.
3 New Genetic Mutations Linked to Male Breast Cancer
Our genes determine everything about our bodies. They play a key role in our health and can determine our likelihood of developing diseases. Some genetic mutations significantly increase the risk of developing breast cancer. The most common mutations associated with breast cancer involve the BRCA1 and BRCA2 genes. About 10% of male breast cancers are caused by mutations in the BRCA2 gene. Other examples of mutations that increase the risk of male breast cancer include those found in PTEN, PALB2, CHEK2 and NBN genes.
In addition to these well-recognized gene mutations, other very minor changes to our DNA code (known as single-nucleotide polymorphisms, or SNPs) also influence our risk of developing cancer. Unfortunately, these types of changes in our genetic code are much more common than BRCA gene mutations.
Although most genetic research associated with breast cancer risk is derived from studies in women, newly published findings bring insight on the genetic risks of developing breast cancer in men. A UK study, published in the Journal of the National Cancer Institute looked at the genotypes of 1,380 men diagnosed with breast cancer.
Results revealed that 3 new SNPs were significantly associated with an increased risk of male breast cancer. One of these SNPs is located on chromosome 6 (rs9371545), and two are located on chromosome 11 (rs554219 and rs78540526). These SNPs increase the risk of developing breast cancer in men by about 47%, 45%, and 61% respectively. Consistent with the predominance of ER-positive tumors in men, genetic correlation was strongest between male breast cancer and ER-positive female breast cancer. Although these genetic mutations are also linked to an increased breast cancer risk in women, the 3 new SNP variants have a greater impact on men.
Improved Decision-Making
While there is still a lot of research still needed, these findings can aid in the development of better risk assessment for men. For those choosing to undergo genetic testing, test results can help quantify individual risk and help guide treatment decision-making around risk-reducing options.
“This study, which shows more of a similarity between the genetic causes of the disease in men and women than previously thought, is a major step forward,” shared Dr Simon Vincent, director of research services at Breast Cancer Now. “Our knowledge of breast cancer in men is limited, because male breast cancer is rare which makes it difficult to collect enough tissue or blood samples to research and understand the disease. We now look forward to further research into the shared genetic causes of male breast cancer which could lead to developing risk reducing treatments and interventions to help prevent more cases among those at increased risk in the future.”
Potential Impact of Delays in Breast Cancer Surgery On Patient Outcomes During COVID-19 Pandemic

The COVID-19 pandemic is impacting many breast cancer patients and their surgical treatment plans across the globe. Many patients today are reporting delays in their breast cancer surgery due to the limitations in place by local governments to help ease the burden on the healthcare system created by COVID-19. The impact of these delays has raised concerns from patients and medical professionals about the long-term impact on patient health. A new study published in the Journal of American College of Surgeons attempts to provide some better understanding.
To gain insight into the effects of surgical delays on early-staged breast cancer patients, the researchers used the National Cancer Database to analyze breast cancer patients treated between 2010 and 2016. About 379,000 patients in the database underwent breast cancer surgery following a diagnosis of DCIS or early-stage (cT1-2N0) ER+ disease. The study evaluated whether longer times between diagnosis and surgical treatment had an impact on 5-year survival rates and cancer staging.
The authors of the study concluded that for women with early-stage breast cancer who had to delay their surgical care due to COVID-19, there should be no impact on overall survival.
The published research also noted there was no negative impact to survival for patients with estrogen sensitive, early-stage breast cancer who were taking tamoxifen or aromatase inhibitors as part of their treatment plan. Interestingly, patients with invasive early-stage breast cancer who had to delay their surgical treatment did not have an increased rate of pathologic upstaging (or a higher cancer stage diagnosis after surgery). Women with ER+ DCIS have a slightly higher risk of upstaging with surgical treatment delays of over 60 days. Similarly, patients with ER- DCIS have a higher risk of upstaging if surgical cancer treatment is delayed more than 120 days. However, even with the elevated risk in upstaging for the DCIS patient populations, there was no impact on their overall survival.
Although these findings cannot accurately account for the delays experienced by patients being treated during the COVID-19 pandemic, this information provides some reassurance for surgeons and their patients dealing with delays in breast cancer surgery.
Does Knowing Your BRCA Status Increase Your Chances of Survival after a Breast Cancer Diagnosis?
Having a BRCA1 or BRCA2 gene mutation increases your risk of developing breast cancer during your lifetime by up to 80%. There are many reasons why you may choose to have or not have genetic testing. However, new research suggests there is a survival benefit associated with knowing that you carry a BRCA1/2 gene mutation.

A recent JAMA study evaluated the connection between patients’ knowledge of their BRCA gene status, and their overall survival after a breast cancer diagnosis. The study analyzed 105 women with breast cancer between 2005 and 2016. Forty-two women knew they were carriers of a BRCA1 or BRCA2 mutation. Sixty-three women only discovered their BRCA status after they were diagnosed with breast cancer. Both groups of women had a similar age at the time of their breast cancer diagnosis. There were no significant differences in hormone receptor status of the tumors in between groups.
Earlier Stage at Diagnosis and Improved Survival
The study found a significant difference between the two groups of women in terms of the breast cancer stage at the time of diagnosis. In the women who knew they carried a BRCA gene mutation, 80.9% had DCIS or a stage 1 cancer at diagnosis, and only 9.5% had a stage 2 breast cancer or higher at diagnosis. In women who did not know their BRCA status, only 30% had an early stage cancer at diagnosis, whereas 52.4% were diagnosed at stage 2 or higher.
‘Knowledge is Power’
Compared to women who knew their BRCA1 or BRCA2 genetic status prior to their breast cancer diagnosis, women who learned their positive gene status after diagnosis were 12 times more likely to have an advanced stage breast cancer at the time of diagnosis. Likewise, 5 year survival was significantly higher for women who knew their BRCA genetic status prior to their breast cancer diagnosis.
Personal Choice
There are pros and cons to every decision, and deciding whether you should have gene testing is no different. There is no “right” or “wrong” and ultimately the decision is a very personal one. Anyone who is considering gene testing should consult with a gene counsellor first to fully discuss the implications of testing ahead of time. There is no point getting a test if you are not prepared to make a decision based on the results.
This study provides some additional helpful information to consider in your decision-making: women who are aware of their BRCA1/2 status have a greater chance of having an earlier stage breast cancer at the time of their cancer diagnosis and living longer, compared to women who carry a BRCA genetic mutation but are unaware.
Managing Breast Cancer Care in the Time of COVID-19

As COVID-19 hotspots continue to appear across the US, it is important for breast cancer patients to know that although some aspects of their treatment plan may continue to be delayed, they are not being forgotten. As more studies about the risks for cancer patients during the pandemic continue to emerge, so has professional medical consensus for safe cancer management and treatment.
The American Society of Breast Surgeons has published recommendations to help guide physicians and their patients through the common scenarios many people now unfortunately found themselves in:
● How do we care for our asymptomatic but high-risk patients presenting for office visits in the post-COVID era?
● How do we handle the backlog of patients whose surgical treatment was delayed due to the pandemic?
● As our operating rooms reopen, how should patients who were placed on endocrine therapy prior to definitive surgery be managed?
● As we emerge from the pandemic, how do we manage patients who have already begun neoadjuvant chemotherapy?
● How do we manage a patient who is not a candidate for breast conserving surgery but is ready for their operation?
It is important for breast cancer patients to understand their treatment plans can be regionally impacted based on local infection rates, available hospital capacity, ICU capacity and the number of available ventilators. Although it can be extremely frustrating, delaying breast cancer surgery is for the safety of patients and will never be delayed if postponement would cause a negative impact on overall survival.
If patients have questions or concerns about regional guidelines as it pertains to breast cancer surgery and/or breast reconstruction, it is very important to reach out to the medical care team directly.
For more information on COVID-19 and breast cancer, continue reading HERE.
COVID-19 Patient Outcomes After Breast Cancer Treatment
Scientists and medical professionals are continuing to learn more about the new Coronavirus strand that has changed our world today. For breast cancer patients, compromised immune systems following treatment place them in the “high risk” category for contracting COVID-19. Likewise, they may also have a higher risk of developing more sever symptoms. However, a recent study sheds a small positive light in the otherwise overwhelming reality of the current pandemic. The study analyzed COVID-19 mortality among patient who had undergone previous breast cancer treatment.
Seventy-six breast cancer patients were enrolled in the study, of which 59 had COVD-19. Of the positive coronavirus enrollees, 37 had metastatic breast cancer, 28 required hospitalization, 6 required admittance to the ICU and 4 died. Results showed COVID-19 deaths were more likely due to other comorbidities rather than current or previous breast cancer treatments. Of those patients who sadly lost their lives, all had significant noncancer comorbidities including hypertension, obesity, diabetes and heart disease.
“While our study cannot determine the incidence of COVID-19 infection among breast cancer patients, the small number of diagnosed cases suggests that breast cancer patients do not appear to be at higher risk than the general population,” the authors of the study share in the findings summary. “Importantly, we found no trend in favor of a relationship between a history of breast and lymph node radiation therapy, radiation therapy sequela, and radiologic extent of disease or outcome.”
Although breast cancer patients need to take every precaution possible to reduce the risk of contracting and spreading COVID-19, if a positive diagnosis does occur, prior breast cancer treatment will likely not impact recovery.
According to the CDC’s guidelines, it is very important we all take the following steps to reduce the risk of contracting and/or spreading COVID-19:
- Wash your hands often
- Avoid close contact with others
- Cover your mouth and nose with a mask or cloth covering when around others, especially indoors
- Cover all coughs and sneezes
- Clean and disinfect surfaces regularly
- Be alert for symptoms and take your temperature regularly
The Financial Burden of Breast Cancer

One in every 8 women in the US will be diagnosed with breast cancer in her lifetime, and 2,000 men will be diagnosed with breast cancer this year. Treatment options, new drugs, surgical advances and breast reconstruction options are all commonly discussed within the breast cancer community. A topic that is not discussed enough is the financial burden, or ‘financial toxicity’ that comes with a breast cancer diagnosis. Financial toxicity is defined as the impact of direct and indirect health care costs that lead to significant financial burden for patients and their caregivers, resulting in increased psychosocial distress, diminished patient outcomes, and a poorer quality of life.
Breast cancer treatment plans can consist of a combination of chemotherapy, radiation, surgery to remove the cancer (lumpectomy or mastectomy), and breast reconstruction. Ideally, patients work closely with their medical team using the process of shared decision-making to determine which plan best fits their individual diagnosis, situation, preferences and goals. However, for many patients, treatment decisions are influenced not by their medical team’s recommendations, but more by the associated costs.
In a published survey of over 600 women, 28% admitted the cost of treatment influenced their treatment decisions. 35% also reported having financial hardships following the completion on their breast cancer treatments. 78% of responders never even discussed costs with their cancer team. These findings likely underestimate the severity of financial toxicity associated with breast cancer due to the lack of diversity within the survey.
A recent analysis of published studies found that few cancer survivors receive financial information and support from healthcare facilities during their initial treatment, despite the widespread impact of cancer-related costs on patients’ health and quality of life.
It should not be assumed that the financial hardship caused following a cancer diagnosis is limited to those without insurance. Even with insurance, high out-of-pocket costs for breast cancer patients can include copayments, deductibles, and coinsurance responsibilities. Cancer treatment can also affect individuals and their care taker’s ability to work, reducing household incomes. For patients who must travel long distances for their cancer care, travel related costs (such as gas, hotel, food, flights) can also add up very quickly.
How can you reduce the financial burden of breast cancer?
1. Discuss costs with your medical team up front.
It is important when discussing treatment options with your healthcare team to be aware of up-front and out-of-pocket costs. Being prepared before beginning treatment can help you plan and budget for the additional costs.
2. Research options that may help you maximize your existing insurance.
Speaking with your HR department or a hospital financial adviser may provide insight to options available to help you get the most out of your insurance, including help with deductibles and co-pays. One such example is the Patient Advocate Foundation.
3. Reach out to non-profit organizations.
There are many national and local breast cancer organizations providing financial assistance to patients (and their families) in need. To find organizations in your area, talk with your medical team and check out the financial assistance tab in the Resources section within the Breast Advocate App – download it here.
Screening Mammograms Shown to Reduce Deaths Caused by Advanced Breast Cancer
There is a constant debate on whether screening mammograms offer a significant benefit to patients.

In the American Cancer Society journal, CANCER, a Swedish study has found that early mammogram screenings can significantly reduce the number of deaths from metastatic breast cancer.
The study evaluated 549,091 women, covering approximately 30% of the screening‐eligible population in Sweden. The results showed that women who had regular screening mammograms had a statistically significant 41% reduction in their risk of dying of breast cancer within 10 years, and a 25% reduction in the rate of advanced breast cancers, compared to individuals who did not participate in mammogram screenings. According to the study, these benefits appeared to be independent of recent changes in patient treatment regimens.
The study concludes that the “benefits of participating in mammography screening are truly substantial and save lives through early detection, lives that otherwise would have been lost under the prevailing therapy at the time of diagnosis.”
This study offers encouraging insight into the benefits of regular breast cancer screening. It is important for both women and men to discuss their overall risk of developing breast cancer to determine the best time to begin screening, and how often they should be screened.
Performing monthly at-home self-breast exams is also a very important, free screening tool everyone should practice in addition to regular radiologic screening. However, this should not replace the imaging screening recommended by your healthcare team.
If access to care or cost is keeping you from following through with your recommended breast cancer screening appointment, please check out the Financial Assistance tab within the Resource section in the Breast Advocate app.
Breast Advocate Founder Joins BRAVE Coalition to Discuss Rescheduling Elective Breast Surgery After COVID-19
Why is there so much confusion on whether elective cases (including breast reconstruction) can start to resume?
Yes, there’s lots of confusion, especially as it relates to a hospital setting versus other surgery centers. Obviously, a lot of a lot of plastic surgeons operate either out of their offices or they have operative suites. Many of them have surgery centers or ambulatory surgical centers. There has been a lot of mixed messaging and some confusion for sure.
For us [in San Antonio], we are operating again. I am very happy with the procedures in place at the local hospitals in terms of testing. I am one of those who believes that we need testing for all patients. Personally, I would not be comfortable offering [breast reconstruction] surgery to people right now without pre-operative COVID-19 testing. There are pros and cons to every approach but that’s just my take.
We must have consistent, good, safe practices and protocols for everyone. Specifically, the surgical community fears operating on asymptomatic positive patients (someone who is positive who doesn’t know it because they’re not showing any symptoms). There’s some data out there that suggests that positive patients who are asymptomatic are at higher risk of developing post-operative complications. You’ve got to take that data seriously.
There was a study out of China that looked at elective patients having surgery (various specialties not just breast reconstruction). They looked at patients who were actually, in retrospect, positive for COVID-19 but at the time hadn’t shown any symptoms so they would have passed all verbal screening measures. Many of those patients ended up getting complications after surgery that otherwise you wouldn’t have expected them to have. There was a very high rate of pneumonia and mortality. Mortality was actually 20%. There’s lots of other data you have to take into account too. What’s the risk of someone being an asymptomatic carrier, what’s the rate of infection in the community…?
In San Antonio, only 10% of people being tested for COVID-19 are coming back positive. But, without mass testing you don’t know what the baseline rate of infection is.
There is a school of thought that’s a lot less restrictive… If you have a low risk patient who hasn’t had any obvious exposure, who passes all the verbal screening and physical screening (no symptoms, normal temperature, normal oxygen saturation on a pulse oximetry)… you don’t need testing on that person. That’s a track that some surgeons are pursuing and there’s definitely some value and an argument to pursue that line, and to reserve testing for only the high-risk group.
I don’t want people to take away from this that if your surgeon doesn’t test you pre-operatively, then you shouldn’t be having the surgery…that’s not what I’m saying. You have to take into account your individual situation, the geographic location, what’s going on where you are, and just have a very honest conversation with your surgeon, and talk about the protocol [they’re using].




