Addressing Disparities between Black and White Women with Triple-Negative Breast Cancer
Multiple studies have shown that African-American women have poorer survival outcomes after a breast cancer diagnosis than white women. One significant contributing factor to these findings is that most studies include all types of breast cancer together. This approach can skew results as white patients have a higher incidence of estrogen receptor-positive breast cancer than black women, which has better outcomes than the more aggressive triple-negative form of the disease. A summary of the different types of breast cancer can be found here.
Survival rates between black and white women with triple-negative breast cancer (TNBC) appear to equalize when these cancers are found early with screening mammograms, a new JAMA study suggests. However, TNBC still remains about twice as common in black women.
This study emphasizes the importance of screening and early detection, particularly in traditionally underserved black women. Unfortunately, we still don’t know why black women experience a much higher rate of TNBC.
What you need to know about Breast Implant Illness (BII)
Breast Implant Illness (BII) is a recently described constellation of symptoms some women have described after receiving a breast implant either for cosmetic augmentation or breast cancer reconstruction.

The US Food and Drug Administration (FDA) notes in public statements and on their website that they have received reports of some women who experienced symptoms with both saline and silicone gel-filled breast implants. These symptoms are termed “systemic” meaning occurring throughout the body and outside of the breast area. The FDA further explains that symptoms are very diverse between patients and may include fatigue, memory loss, rash, “brain fog,” and joint pain.
Patients and some caregivers have used the term “breast implant illness” (BII) to describe some or all of these symptoms. These symptoms may sometimes develop after only a few months or many years after receiving the implants.
Currently, these symptoms and what causes them are poorly understood and there are no known blood tests or imaging studies to test for or confirm BII. Even when women display these symptoms, blood tests are frequently normal and do not demonstrate any infection or inflammation.
There are no known factors that have been identified that increase risk of developing these symptoms. BII symptoms appear to only affect a small minority of patients receiving breast implants. The FDA notes that in some cases, removal of the breast implants without replacement has been reported to reverse symptoms of breast implant illness. Importantly, the FDA has not detected any association between breast implants and breast cancer or reproductive problems. Currently, researchers are investigating these symptoms to better characterize BII, understand the origins, and relation to breast implants.
For any patients that may experience these symptoms or for any injury with a medical device, the FDA encourages reporting by phone at 1-800-FDA-1088 or online at MedWatch, the FDA Safety Information and Adverse Event Reporting program.
About the author:
Dr Mark Clemens is a board certified plastic and reconstructive surgeon at the MD Anderson Cancer Center, and contributor to the Breast Advocate® App. Dr Clemens’ research interests include outcomes and translational research involving microvascular and prosthetic reconstructive surgery of the breast. He is also a leading authority on breast implant-associated anaplastic large cell lymphoma (bia-ALCL) and co-leads a multidisciplinary clinical and laboratory research team on the investigation of bia-ALCL.
WHO Increases Worldwide Access To Life-saving Breast Cancer Treatment
Last month the World Health Organization (WHO) announced the approval of a “biosimilar” Trastuzumab to help make breast cancer treatment more affordable for patients globally.
Biosimilar drugs are derived from living sources rather than chemicals. Biosimilars are approved according to the same pharmaceutical quality standards that apply to all medicines.
The WHO shared in their statement that the drug Trastuzumab, a monoclonal antibody, is an essential treatment for about 20% of breast cancers. It has shown high efficacy in curing early stage breast cancer and even some cases of more advanced disease. However, the average cost of the drug is around $20,000 a year making it difficult for most patients around the world to afford the treatment. The WHO-approved biosimilar version of the drug has the same effectiveness but costs about 65% less. This major cost difference gives hope to many seeking affordable access to treatment. Previously, other biosimilar versions of Trastuzumab have come to market, but none have passed qualification standards set forth by the WHO.
“WHO prequalification of biosimilar trastuzumab is good news for women everywhere,” said WHO director general Dr. Tedros Adhanom Ghebreyesus. “Effective, affordable breast cancer treatment should be a right for all women, not the privilege of a few,” he added.
“We need to act now and try to avoid more preventable deaths,” said WHO assistant director general for medicines and health products Dr Mariângela Simão. “The availability of biosimilars has decreased prices, making even innovative treatments more affordable and hopefully available to more people.”
Noteworthy Highlights from SABCS 2019
Breast cancer research continues to expand experts’ knowledge and understanding of the disease and how to treat it. Each year healthcare experts from across the globe gather in San Antonio to present the latest research findings at the San Antonio Breast Cancer Symposium (SABCS). Here is a great video overview of some highlights from this year’s meeting:
For more videos discussing specific studies presented at SABCS 2019, please click here.
Can Frequently Coloring or Straightening Your Hair Increase your Risk of Breast Cancer?
A recent study published in the International Journal of Cancer identifies a potential link between permanent hair dye and chemical hair straighteners with a higher risk of breast cancer. The study followed 46,709 women whose sisters had previously been diagnosed with breast cancer.
The results showed that the women who used permanent hair dye or straighteners (or applied straighteners to others) within the year prior to enrolling in the study were 9% more likely to develop breast cancer compared to the women who did not use these products. This association was even stronger in black women who showed a 45% higher risk of developing the disease; this is consistent with toxicology reports that have found higher concentrations of estrogens and hormone-disrupting chemicals in hair products marketed to black women. Women who only used temporary or semi-permanent dyes showed no increased risk for breast cancer.
Permanent hair dyes and chemical straighteners are used by many women and therefore represent a very prevalent exposure. This study suggests that frequent use of these products could increase the risk of breast cancer, particularly in women with a family history of the disease. There are also significant differences in risk based on ethnicity since products marketed to black women contain the highest amount of potentially carcinogenic chemicals.
Supplemental MRI Screening May Benefit Women with Extremely Dense Breast Tissue
Women with extremely dense breast tissue have a 4 to 6 times increased risk of developing breast cancer, and their cancers are also less likely to be detected on a mammogram. About 50% of women have dense breasts.
New laws in many US states were recently passed requiring mammogram reports to include information on the density of a woman’s breast tissue. Dense breast tissue is a risk factor for breast cancer and can make detecting breast cancer more difficult with screening mammograms alone. For this reason, more research is underway to determine how to best screen for cancer in women with dense breasts.
So what are “dense breasts”? Breasts consist of fibrous glandular tissue and fat. Dense breasts contain more fibrous tissue and less fat. On a mammogram, dense fibrous tissue has the same white appearance as a breast cancer. This can make it very difficult for radiologists to spot the breast cancer.
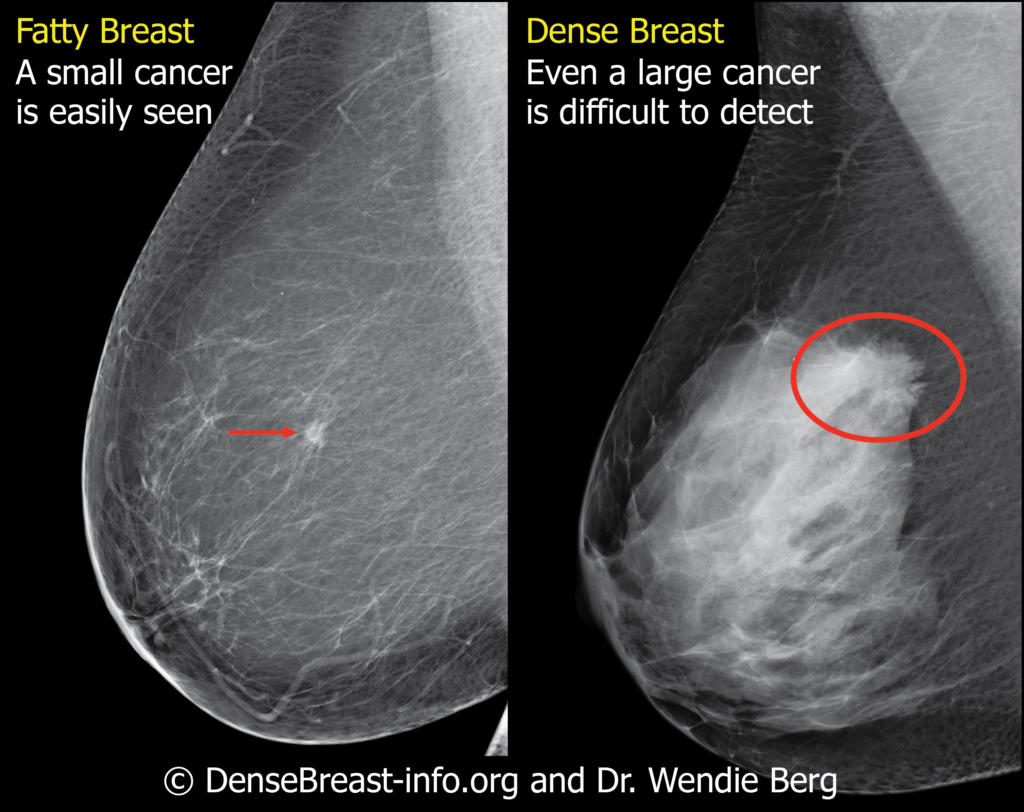
A long-standing question is whether or not women with dense breasts should undergo additional imaging screening tests in combination with mammograms. A new study published in the New England Journal of Medicine offers new insight supporting the addition of MRI screening to mammograms for women with extremely dense breast tissue.
The study was conducted in the Netherlands. 40,373 women between the ages of 50 and 75 years with extremely dense breasts and normal screening mammograms were randomized to undergo additional screening via MRI, or receive no additional screening. The study results showed the mammogram plus MRI group experienced 50% fewer interval cancers than the mammogram-only group during a 2-year period (2.5 vs 5 per 1,000 screenings, respectively). The tumors detected on MRI were smaller, of an earlier stage, and more likely to be node-negative than those detected in the mammography-only group.
This study suggests that a combination of mammograms and MRIs can be beneficial for women with extremely dense breast tissue in detecting breast cancer at an earlier stage than mammograms alone. However, it is important to note the results of this study do not suggest adding MRIs will decrease the death rates from breast cancer.
It is important to discuss your screening options with your health care team to see if additional MRI imaging would be beneficial for you based on your specific situation and risk of developing breast cancer.
Weight-loss Surgery May Decrease Breast Cancer Risk
According to US Centers for Disease Control and Prevention (CDD), obesity is associated with an increased risk for 13 types of cancer, including breast cancer.
For individuals at high risk for developing breast cancer, maintaining a healthy lifestyle and BMI is important for reducing the likelihood of developing the disease. In some situations, weight loss surgery may be a good option to help reduce the overall risk.
According to a new study presented by Cleveland Clinic Florida researchers at the 36th American Society for Metabolic and Bariatric Surgery (ASMBS) Annual Meeting at ObesityWeek 2019, weight loss surgery cut the overall risk of developing cancers linked to obesity by 20%. In fact, women with obesity and known genetic susceptibilities for breast cancer were 2.5 times more likely to develop breast cancer than women with the same genetic risk who underwent weight loss surgery.
“Our findings suggest bariatric surgery could significantly prevent the development of cancer in patients with a higher risk than the average population, even in those genetically predisposed,” said study co-author Emanuele Lo Menzo, MD, Ph.D., FASMBS, Associate Program Director, General Surgery Residency Program, Cleveland Clinic Florida in Weston. “The effect we saw on patients genetically predisposed to developing breast cancer was remarkable and we believe this is the first time a study has shown such an impact. Further studies are needed to determine the factors, including weight loss, that may have led to such risk reduction.”
It is interesting to note that a similar study of patients with severe obesity (BMI of 35 or higher) published in Annals of Surgery earlier this year showed weight-loss surgery was associated with a 33% decrease in the risk of developing any type of cancer, and a 40% decrease in the risk of being diagnosed with a cancer associated with obesity.
It is important to remember, even individuals with a healthy BMI are still at risk for developing cancer. The importance of achieving and maintaining a healthy weight is to reduce the likelihood of developing breast cancer – unfortunately, it is NOT guaranteed prevention.
If you struggle to maintain a healthy weight, you are not alone! In fact, maintaining a healthy weight is a challenge for most adults today. The CDC reports more than 70% of American adults are overweight.
Maintaining a healthy weight is also very important after breast cancer treatment to decrease the risk of a recurrence. However, add the impact of a breast cancer diagnosis and the side-effects of some treatments, losing weight after breast cancer is typically even harder. Weight loss surgery may therefore also be an option for some survivors to consider if all other options have been exhausted.
Most people know that calorie control and regular exercise are crucial in losing and maintaining weight, but few people realize 80% of weight loss is achieved through better food choices. Even when we think we’re doing well by choosing the salad, we don’t realize the dressing has more calories than a Big Mac! A consultation with a nutritionist or dietician is a very good place to start your weight loss journey, and can provide extremely useful guidance in planning healthier meals.
Staging Nipple-Sparing Mastectomy can Decrease the Risk of Complications
Nipple-sparing mastectomy (NSM) is the latest evolution in mastectomy technique. The procedure preserves the entire skin envelope and nipple-areola. Only the underlying breast tissue is removed. Nipple-sparing mastectomy significantly improves cosmetic results when combined with immediate breast reconstruction, and is oncologically safe when performed in appropriate candidates. It can also improve the return of sensation in some patients.
However, certain factors such as large breast size and a low nipple location can increase the risk of complications such as partial or even complete necrosis of the nipple and areola. There is some good news though for patients choosing NSM to decrease their risk of getting breast cancer (“prophylactic” or “risk-reducing” NSM)… “Staged” surgery with a breast reduction performed at least three months before the NSM significantly decreases these complications in patients with larger breasts.
According to a study published in Plastic and Reconstructive Surgery that compared the staged approach with the traditional all-in-one surgery, staged surgery patients experienced no major skin or nipple-areola necrosis. However, patients who did not reduce their breast size via a breast reduction before having their NSM and immediate reconstruction experienced major necrosis 22% of the time.
The staged approach allows surgeons to reduce the patient’s overall breast size, remove excess skin, and reposition the nipple-areola before the definitive NSM and reconstruction. By decreasing the overall size of the breast and relocating the nipple-areola to a more favorable position ahead of time, the demands on the blood supply at the time of the NSM are significantly reduced. This in turn decreases the risk of complications.
Staging does add an addition surgery and cost, along with a longer overall recovery period, but should be considered in patients who may otherwise not be good candidates for risk-reducing NSM due to large breast size or a low-lying nipple-areola. Patients with a cancer diagnosis are usually not candidates for this staged approach due to the delay it adds to treatment of the cancer.
Free App for Breast Cancer Treatment and Breast Reconstruction Decision-Making
A leader in breast cancer treatment education and shared decision-making, Breast Advocate® is excited to announce the release of our updated free app for breast cancer surgery and breast reconstruction. Thank you to our users for providing the valuable feedback that has helped make our app even better!

The latest updates greatly improve user experience and navigation, and give the innovative breast cancer app a fresh new look. Users can now customize the content they see on their dashboard via a personal feed, and easily find specific content using our optimized search engine.
Co-created by breast cancer specialists and patient advocates, the Breast Advocate® app provides anyone with breast cancer, a history of breast cancer, or at high risk for developing the disease a much needed voice in their treatment decision-making.
Should you have a lumpectomy or mastectomy? Should you have prophylactic surgery? Should you go flat or have breast reconstruction? If you want reconstruction, what type is best for you? Should you have gene testing? Our breast cancer app can help with these questions and much more.
The groundbreaking Breast Advocate® Wizard walks users through a detailed questionnaire to better understand their personal situation, preferences and goals. Our proprietary software combines evidence-based treatment options with the user’s preferences to create customized recommendations that are in line with the user’s specific needs. Breast Advocate® empowers users with the knowledge they need to fully discuss their treatment plan with their medical team and ensure they make the best decisions for them.
Users also have access to the latest expert opinions, published studies, helpful resources, and a community ready to provide support.
Download the latest version of the Breast Advocate app here.
Are Breast Cancer Risk-Reducing Medications Right For You?
With the exception of skin cancer, breast cancer is the most common cancer among American women, and the second leading cause of cancer deaths in the US. Women with a high risk of developing breast cancer have the option of lowering their risk by taking certain medications.
The US Preventive Services Task Force (USPSTF) recently made two updates to their recommendations for risk-reduction medications:
“The USPSTF recommends that clinicians offer to prescribe risk-reducing medications, such as tamoxifen, raloxifene, or aromatase inhibitors, to women who are at increased risk for breast cancer and at low risk for adverse medication effects”. This recommendation is for women who have never been diagnosed with breast cancer, but have at least a 3% risk of developing the disease within 5 years.
“The USPSTF recommends against the routine use of risk-reducing medications, such as tamoxifen, raloxifene, or aromatase inhibitors, in women who are not at increased risk for breast cancer… This recommendation applies to asymptomatic women 35 years and older, including women with previous benign breast lesions on biopsy such as atypical ductal hyperplasia or lobular hyperplasia, and LCIS. This recommendation does not apply to women who have a current or previous diagnosis of breast cancer or ductal carcinoma in situ.” In this group of patients, the side effects associated with taking these medications would likely outweigh the prevention benefit.
The USPSTF based its recommendations on the evidence of the benefits and potential side effects of taking any of these risk-reducing medications, and an assessment of the balance. The USPSTF does not consider the cost of care in any of their assessments.
The USPSTF also states that it recognizes that clinical decisions involve more considerations than evidence alone. We strongly encourage a shared decision-making approach between patients and their healthcare team to determine what’s best for you. If you would like to learn if you could benefit from taking a risk-reducing medication and whether taking one is right for you, please discuss the full range of options, risks and benefits with your physician.
You can read the full USPSTF statement here.




