FDA Calls For Men To Be Included In Breast Cancer Clinical Trials
Although breast cancer in men is not often publicized in the media, it is very important to know that men are at risk of the disease too. It is estimated that about 2,600 men will be diagnosed and 500 will die of breast cancer each year. However, because men contribute less than 1% of all cases, they are typically not included in clinical trials for treatment.
Since men have not historically been included in clinical trials, it is very possible they are not receiving optimal care. The drugs and treatment options they are prescribed have only been tested and approved for women.
The Food and Drug Administration (FDA) is calling for this to change. In their recent draft guidance, the FDA recommends the inclusion of men in breast cancer clinical trials to help improve the clinical management of the disease in male breast cancer. The FDA also states that if researchers propose to exclude male patients from a breast cancer trial, “scientific rationale should be included.” Furthermore, the FDA will not consider “low expected accrual rates of male patients with breast cancer” as sufficient rationale for excluding male patients moving forward.
The move seems to be working already… Following the FDA’s announcement, some breast cancer drug studies have extended eligible enrollment criteria to men.
USPSTF Expands BRCA Gene Testing But Not Far Enough
The US Preventive Services Task Force (USPSTF) issued a new recommendation statement calling for more patients to receive genetic counseling and genetic testing for BRCA1 and BRCA2 gene mutations.
Often referred to as the “breast cancer gene,” mutations in BRCA1 and BRCA2 genes have been shown to increase the likelihood of an individual developing breast, ovarian, fallopian tube and peritoneal cancer.
To better predict and manage the risk of developing hereditary cancers, the USPSTF is now recommending more patients, including breast, ovarian, fallopian tube and peritoneal cancer survivors, undergo genetic counseling and genetic testing. Previously, the USPSTF only recommended those who had a family history BRCA-related cancers be tested.
The USPSTF statement, published in the JAMA Network, recommends that “primary care clinicians assess women with a personal or family history of breast, ovarian, tubal, or peritoneal cancer or who have an ancestry associated with BRCA1/2 gene mutations with an appropriate brief familial risk assessment tool. Women with a positive result on the risk assessment tool should receive genetic counseling and, if indicated after counseling, genetic testing… The USPSTF recommends against routine risk assessment, genetic counseling, or genetic testing for women whose personal or family history or ancestry is not associated with potentially harmful BRCA1/2 gene mutations.”
Why are these tests important? Expanding testing will potentially help survivors determine which treatments would be best based on their current diagnosis, as well as alert them to treatments that could significantly lower their future cancer risk, such as risk-reducing mastectomy and hysterectomy. It could also be very important and possibly life-saving for some of the survivors’ family members.
While the updated recommendations are very good news, in several ways they do not go far enough: newly diagnosed patients with breast or ovarian cancer, patients with advanced cancers, and men were not included. Men who carry a BRCA gene mutation are at increased risk for breast, pancreatic and prostate cancers, and should also have counseling and testing. As in women, these mutations can be passed on to their children and beyond.
Anyone who is concerned about their future cancer risk and doesn’t fall into these guidelines should consider proactively seeking a consultation with a genetic counselor. You can find a certified genetic counselor near you via the National Society of Genetic Counselors.
New Blood Test could predict Breast Cancer Recurrence and even prevent Surgery
While there is still much work to be done, detection and treatment of early stage breast cancer have improved tremendously over the last few years. Chemotherapy regimens have become increasingly effective, boosted by the emergence of immunotherapy, and often lead to a ‘pathological complete response’ when given before surgery (known as ‘neoadjuvant therapy’). A pathologic complete response means there is no identifiable tumor in the tissue that is resected by the lumpectomy or mastectomy.
However, there is currently no way to truly know which patients treated for early stage breast cancer enter remission and which ones actually have residual cancer cells in their system (in the absence of obvious metastatic disease).
A recent study published in Science Translational Medicine reported very encouraging news on a test called Targeted Digital Sequencing (or ‘TARDIS’) that could potentially help predict which patients are at high risk of breast cancer recurrence. TARDIS identifies DNA released by breast cancer cells into the bloodstream but is up to 100 times more sensitive than other liquid-biopsy tests currently available or in development.
This could be a huge step forward since TARDIS will potentially help doctors identify patients with early stage breast cancer who may still have residual cancer cells in their bodies after treatment that aren’t otherwise detectable by standard scans and follow-up monitoring.
It is also possible that the test could help remove the need for surgery altogether in women who experience a pathological complete response with neoadjuvant chemotherapy and are shown to have no residual breast cancer DNA in their bodies.
Very exciting news indeed! Read the full article here.
Allergan Recalls its Natrelle BIOCELL Textured Breast Implants and Tissue Expanders due to risk of BIA-ALCL
On July 24th, Allergan announced it was recalling its BIOCELL textured expanders and implants from the global market in response to a request from the FDA. The FDA made the request that Allergan recall all its BIOCELL textured breast implants and tissue expanders based on newly submitted Medical Device Reports reporting worldwide cases of Breast Implant Associated Anaplastic Large-Cell Lymphoma (BIA-ALCL), and related deaths associated with these devices.
BIA-ALCL is a rare cancer of the immune system that develops in the scar tissue surrounding breast implants. Common symptoms include swelling, significant fluid accumulation around the implant, pain and/or lumps in the breast. When caught early, the cancer is curable by removing the breast implant and the surrounding scar tissue (known as a capsulectomy). Patients with advanced disease need more treatment in the form of chemotherapy and radiation.
To date, 573 cases of BIA-ALCL and 33 deaths have been confirmed worldwide. Additional cases have been reported but have not been confirmed. Of the 573 confirmed cases, 481 have been attributed directly to Allergan’s Biocell textured implants leading to a global recall of the devices.
The US recall comes after an official FDA hearing was held in March to evaluate textured breast implant safety. The FDA first reported the possible link between breast implants and anaplastic large cell lymphoma in 2011. All confirmed cases have been attributed to textured implants. While there have also been cases in women who had smooth implants, due to the lack of a comprehensive medical history and incomplete implant information in these patients, there have been no confirmed cases attributed solely to smooth implants.
Although the Allergan textured implants have been recalled, the FDA is not recommending preventative removal of these textured implants in women who don’t have any symptoms due to the very low risk of developing BIA-ALCL. The FDA does recommend patients be educated on the potential risks associated with textured implants as well as the signs and symptoms of any complications. Patients are encouraged to contact their plastic surgeons with any questions or concerns whatsoever about their implants.
Read the full FDA patient safety communication here.
Will this detect breast cancer 5 yrs earlier?
MIT’s Computer Science and Artificial Intelligence Lab has developed a new tool that uses artificial intelligence (AI) and deep learning to predict the development of breast cancer up to five years earlier than current detection techniques.
Unlike other AI-based tools that use data mostly from acquired from white patients and may therefore be algorithmically biased, MIT’s model works equally well for both white and black patient populations. This is particularly important as black women are 42% more likely to die from breast cancer than white women – a possible contributing factor could be that current detection techniques don’t work as well in black women.

To create the tool, data was obtained from over 90,000 mammograms and outcomes from over 60,000 patients treated at the Massachusetts General Hospital. A form of machine learning known as “deep learning” was then used to identify patterns in the images and data that are too subtle to be routinely recognized by human physicians. The results have so far have been more accurate than current diagnostic approaches, presumably because the model is not based on existing knowledge and assumptions about patient risk factors.
MIT’s project is intended to help doctors compile the best personalized screening program for their patients and hopefully eliminate the heartbreaking outcome of a late breast cancer diagnosis. We eagerly await to hear if and when this could become widely available.
New surgical guideline leads to lower re-operation rates after lumpectomy surgery
Re-operation rates following breast conservation (lumpectomy and radiation) for early invasive breast cancer have been highly variable historically, mainly because of uncertainty and variability in what surgeons deemed safe.
When a tumor is removed, the specimen is painted with a special ink before it is evaluated by a pathologist. This ink allows the pathologist to clearly see the outer edges, or ‘margins’, of the tissue under the microscope.
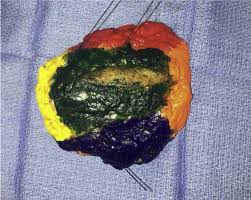
In 2014, the Society of Surgical Oncology and American Society for Radiation Oncology released new guidelines stating that as long as the tumor has no ink on it, the margin is clear. A clear margin means there are no cancer cells at the edge of the surgical specimen and tells the surgeon that all the cancer was removed.
Prior to the new guidelines, some surgeons wanted at least 2mm of normal breast tissue around the cancer. Others accepted less. Even though wider clear margins don’t reduce the risk of cancer recurrence, because of this lack of consensus, 25%-30% of patients having a lumpectomy required more surgery to ensure a larger clear margin.
A recent study in the American Journal of Surgery shows that the new guidelines have led to a decrease in re-operation rates. This offers peace of mind that you likely won’t need another “re-excision” surgery after a lumpectomy if the margins are clear, irrespective of how small the clear margins are, as long as there is “no ink”. Additionally, surgeons now have an evidence-based standard to follow. Very reassuring news indeed!
Does DIEP flap breast reconstruction increase the risk of breast cancer recurrence?
Patients choosing to undergo breast reconstruction are often concerned that their decision may increase their risk of breast cancer recurrence. A recent study published in the British Journal of Surgery looked at the risk of recurrence specifically after DIEP flap reconstruction.
The study compared 250 patients who had DIEP flap reconstruction between 1999 and 2013 with more than 700 control patients. Breast cancer recurrence was seen in over 19 percent of patients who had DIEP flap surgery and over 23 percent of patients in the control group. Interestingly, the 5-year breast cancer-specific survival rate was highest in patients having DIEP flap surgery after 2008 – 92.4 percent in the DIEP flap group versus 87.4 percent in the control group.
The study design does raise some questions about selection bias. For example, all DIEP flap procedures in this study were performed at least 24 months after mastectomy. No DIEP flaps were performed at the same time as the mastectomy (‘immediate’ reconstruction). In addition, a larger number of patients in the DIEP flap group than the control group received adjuvant therapy.
While the survival rates between groups may have been influenced by patient selection, this study shows that patients with breast cancer undergoing DIEP flap breast reconstruction do NOT have a higher rate of breast cancer recurrence than patients undergoing mastectomy alone.
Breast Advocate Founder Presents at Male Breast Cancer Coalition’s Annual Conference
According to the American Cancer Society, about 2,670 new cases of invasive male breast cancer will be diagnosed in 2019 and about 500 men will die because of this diagnosis.
Male breast cancer is rarely discussed in breast cancer awareness campaigns. The Male Breast Cancer Coalition (MBCC) aims to change this. Their mission is to build awareness of male breast cancer through the wisdom and experiences of survivors through speaking arrangements, social media and their annual conference.
This year, Breast Advocate founder, Dr. Minas Chrysopoulo, was honored to join the MBCC at their annual conference last weekend to discuss male breast reconstruction options. “Like women, men can experience the same concerns about their appearance following breast cancer surgery, most commonly a mastectomy,” shares Dr. Chrysopoulo. “Knowing there are reconstructive options is a great relief to some male breast cancer patients.”
Male breast reconstruction can include several different procedures…
Fat grafting is the most commonly performed method of reconstruction in male breast cancer patients. The procedure is performed by removing fat (using liposuction) from one area of the body, purifying it, and re-injecting it into the chest to fill in chest contour deformities.
A small breast implant can be used to reconstruct a male breast after a mastectomy. Since the aesthetics of the male breast are usually much different to that of woman, a custom implant is often the best choice for the best symmetry.
Some patients can have extensive chest wall deformity following breast cancer treatment, especially if part of the pectoral muscle is removed because of tumor involvement. In these situations, an autologous flap (reconstruction using the patient’s own tissue) may be a good option. Flap options include a Latissimus Dorsi (or “lat”) flap which uses (part or all) of the back muscle below the shoulder blade along with a segment of overlying fat and skin, or the DIEP flap which uses skin and fat from the lower abdomen.
Nipple reconstruction can be performed on its own or in conjunction with any other reconstructive procedure. If desired, the reconstructed nipple and areola can later be tattooed for a more natural appearance.
- Scar Revision Surgery
Scar revision can improve the appearance of breast surgery scars after mastectomy or lumpectomy and can be used in conjunction with fat grafting to improve chest contour defects. Releasing tethered scars can also help reduce discomfort.
- Tattooing
Various forms of tattooing can be performed along with other reconstructive procedures. These include:
- A nipple-areola tattoo (2D or 3D), either alone or in conjunction with nipple reconstruction
- An ornamental tattoo covering the surgical site
Male breast reconstruction (regardless of the procedure) is usually performed after completion of all breast cancer treatment. In select cases, it can be performed at the same time as the mastectomy.
Why you Need to Know Your Breast Density
A recently passed law means that for the first time in over 20 years, the FDA is updating regulations for mammography screening to give patients more information about their breasts. The amendments will specifically require that patients be informed of their breast density in all U.S. states.
Breast tissue that is more fatty in consistency appears dark gray or even black on a mammogram, which makes it fairly easy to identify a breast cancer since that appears as a white lesion on mammograms. Dense breast tissue consists mostly of glandular tissue and is not fatty at all. Glandular tissue also appears white on a mammogram making it harder to detect a cancer. Dense breasts can therefore mask a breast cancer.
Younger women are much more likely to have dense breasts than older women. As women approach menopause, the glandular tissue is gradually replaced by more fatty tissue.
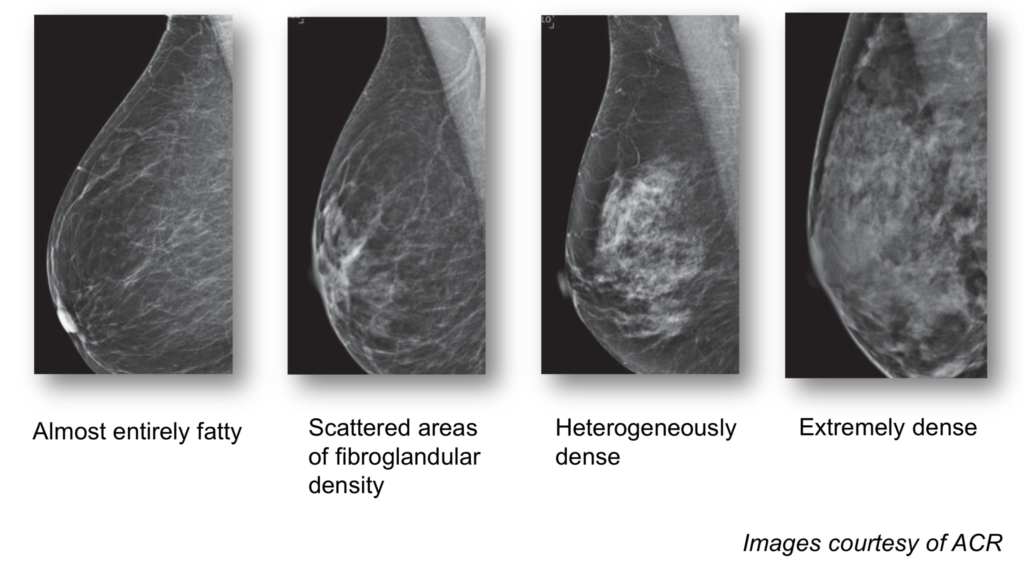
About 50% of women in the U.S. have dense breasts. Women with the densest breasts are up to six times more likely to develop breast cancer than women with fatty breasts. Once diagnosed, a woman with dense breasts may have an increased chance of developing aggressive or metastatic breast cancer, which decreases her overall chance of survival.
The FDA will now require mammography centers and radiologists to share the patient’s breast density information on the mammogram report, in easy understandable language, along with how it might influence the accuracy of the mammogram. Women with dense breasts have additional screening options including 3D mammography, breast ultrasound, and MRI.
The new law is a major victory for the patient advocacy movement, in particular AreYouDense.org, founded by the late Nancy Capello, PhD. Dr Capello was responsible for the first state-based density reporting law passed in Connecticut in 2009 and spearheaded efforts to standardize the communication of dense breast tissue nationally through changes in the Mammography Quality Standards Act.
Reports of Breast Implant-Related Illnesses Prompt FDA Review
New safety concerns over breast implants has prompted the FDA to hold a two-day meeting to hear from researchers, patients, plastic surgeons, and implant manufacturers. The hearing was scheduled for March 25th and 26th, 2019.
Concerns surfaced recently following the publication of the first study to review breast implant safety data following the FDA’s approval of silicone implants. The study, published in Annals of Surgery, is the largest ever study of silicone breast implants.
The researchers analyzed data on nearly 100,000 patients enrolled in the study between 2007 and 2010. More than 80,000 patients received silicone implants (Allergan or Mentor); the rest received saline implants. Seventy-two percent of the patients underwent primary breast augmentation, 15 percent had revision augmentation, 10 percent had primary breast reconstruction, and three percent had revision reconstruction procedures.
The authors found silicone implants are generally safe but are also associated with a slightly higher risk of a few rare diseases including Sjogren’s syndrome (8-fold increase), scleroderma (7-fold increase), rheumatoid arthritis (6-fold increase), stillbirth (4.5-fold increase), and melanoma (4-fold increase). One case of BI-ALCL (rare lymphoma associated with textured implants) was reported.
The findings have been disputed by many experts, including federal health regulators, who nevertheless scheduled the public meeting to consider implant safety.
“These findings aren’t meant to suggest implants caused these problems,” said Dr. Mark Clemens, an MD Anderson professor of plastic surgery, Breast Advocate App contributor, and the senior investigator of the study. “They’re associations, which underscore the need for more research in this area, but do provide key safety information for women and their providers to consider when thinking about cosmetic or reconstructive surgery with breast implants.”
Since the publication, the FDA has also issued a statement acknowledging that implantable devices, including breast implants, may make some people sick. “A growing body of evidence suggests that a small number of patients may have biological responses to certain types of materials in implantable or insertable devices… [which can include] inflammatory reactions and tissue changes causing pain and other symptoms that may interfere with their quality of life.”
Following the hearing and consideration of all the information presented, the FDA released this statement.




