Weight-loss Surgery May Decrease Breast Cancer Risk
According to US Centers for Disease Control and Prevention (CDD), obesity is associated with an increased risk for 13 types of cancer, including breast cancer.
For individuals at high risk for developing breast cancer, maintaining a healthy lifestyle and BMI is important for reducing the likelihood of developing the disease. In some situations, weight loss surgery may be a good option to help reduce the overall risk.
According to a new study presented by Cleveland Clinic Florida researchers at the 36th American Society for Metabolic and Bariatric Surgery (ASMBS) Annual Meeting at ObesityWeek 2019, weight loss surgery cut the overall risk of developing cancers linked to obesity by 20%. In fact, women with obesity and known genetic susceptibilities for breast cancer were 2.5 times more likely to develop breast cancer than women with the same genetic risk who underwent weight loss surgery.
“Our findings suggest bariatric surgery could significantly prevent the development of cancer in patients with a higher risk than the average population, even in those genetically predisposed,” said study co-author Emanuele Lo Menzo, MD, Ph.D., FASMBS, Associate Program Director, General Surgery Residency Program, Cleveland Clinic Florida in Weston. “The effect we saw on patients genetically predisposed to developing breast cancer was remarkable and we believe this is the first time a study has shown such an impact. Further studies are needed to determine the factors, including weight loss, that may have led to such risk reduction.”
It is interesting to note that a similar study of patients with severe obesity (BMI of 35 or higher) published in Annals of Surgery earlier this year showed weight-loss surgery was associated with a 33% decrease in the risk of developing any type of cancer, and a 40% decrease in the risk of being diagnosed with a cancer associated with obesity.
It is important to remember, even individuals with a healthy BMI are still at risk for developing cancer. The importance of achieving and maintaining a healthy weight is to reduce the likelihood of developing breast cancer – unfortunately, it is NOT guaranteed prevention.
If you struggle to maintain a healthy weight, you are not alone! In fact, maintaining a healthy weight is a challenge for most adults today. The CDC reports more than 70% of American adults are overweight.
Maintaining a healthy weight is also very important after breast cancer treatment to decrease the risk of a recurrence. However, add the impact of a breast cancer diagnosis and the side-effects of some treatments, losing weight after breast cancer is typically even harder. Weight loss surgery may therefore also be an option for some survivors to consider if all other options have been exhausted.
Most people know that calorie control and regular exercise are crucial in losing and maintaining weight, but few people realize 80% of weight loss is achieved through better food choices. Even when we think we’re doing well by choosing the salad, we don’t realize the dressing has more calories than a Big Mac! A consultation with a nutritionist or dietician is a very good place to start your weight loss journey, and can provide extremely useful guidance in planning healthier meals.
Staging Nipple-Sparing Mastectomy can Decrease the Risk of Complications
Nipple-sparing mastectomy (NSM) is the latest evolution in mastectomy technique. The procedure preserves the entire skin envelope and nipple-areola. Only the underlying breast tissue is removed. Nipple-sparing mastectomy significantly improves cosmetic results when combined with immediate breast reconstruction, and is oncologically safe when performed in appropriate candidates. It can also improve the return of sensation in some patients.
However, certain factors such as large breast size and a low nipple location can increase the risk of complications such as partial or even complete necrosis of the nipple and areola. There is some good news though for patients choosing NSM to decrease their risk of getting breast cancer (“prophylactic” or “risk-reducing” NSM)… “Staged” surgery with a breast reduction performed at least three months before the NSM significantly decreases these complications in patients with larger breasts.
According to a study published in Plastic and Reconstructive Surgery that compared the staged approach with the traditional all-in-one surgery, staged surgery patients experienced no major skin or nipple-areola necrosis. However, patients who did not reduce their breast size via a breast reduction before having their NSM and immediate reconstruction experienced major necrosis 22% of the time.
The staged approach allows surgeons to reduce the patient’s overall breast size, remove excess skin, and reposition the nipple-areola before the definitive NSM and reconstruction. By decreasing the overall size of the breast and relocating the nipple-areola to a more favorable position ahead of time, the demands on the blood supply at the time of the NSM are significantly reduced. This in turn decreases the risk of complications.
Staging does add an addition surgery and cost, along with a longer overall recovery period, but should be considered in patients who may otherwise not be good candidates for risk-reducing NSM due to large breast size or a low-lying nipple-areola. Patients with a cancer diagnosis are usually not candidates for this staged approach due to the delay it adds to treatment of the cancer.
Free App for Breast Cancer Treatment and Breast Reconstruction Decision-Making
A leader in breast cancer treatment education and shared decision-making, Breast Advocate® is excited to announce the release of our updated free app for breast cancer surgery and breast reconstruction. Thank you to our users for providing the valuable feedback that has helped make our app even better!

The latest updates greatly improve user experience and navigation, and give the innovative breast cancer app a fresh new look. Users can now customize the content they see on their dashboard via a personal feed, and easily find specific content using our optimized search engine.
Co-created by breast cancer specialists and patient advocates, the Breast Advocate® app provides anyone with breast cancer, a history of breast cancer, or at high risk for developing the disease a much needed voice in their treatment decision-making.
Should you have a lumpectomy or mastectomy? Should you have prophylactic surgery? Should you go flat or have breast reconstruction? If you want reconstruction, what type is best for you? Should you have gene testing? Our breast cancer app can help with these questions and much more.
The groundbreaking Breast Advocate® Wizard walks users through a detailed questionnaire to better understand their personal situation, preferences and goals. Our proprietary software combines evidence-based treatment options with the user’s preferences to create customized recommendations that are in line with the user’s specific needs. Breast Advocate® empowers users with the knowledge they need to fully discuss their treatment plan with their medical team and ensure they make the best decisions for them.
Users also have access to the latest expert opinions, published studies, helpful resources, and a community ready to provide support.
Download the latest version of the Breast Advocate app here.
Are Breast Cancer Risk-Reducing Medications Right For You?
With the exception of skin cancer, breast cancer is the most common cancer among American women, and the second leading cause of cancer deaths in the US. Women with a high risk of developing breast cancer have the option of lowering their risk by taking certain medications.
The US Preventive Services Task Force (USPSTF) recently made two updates to their recommendations for risk-reduction medications:
“The USPSTF recommends that clinicians offer to prescribe risk-reducing medications, such as tamoxifen, raloxifene, or aromatase inhibitors, to women who are at increased risk for breast cancer and at low risk for adverse medication effects”. This recommendation is for women who have never been diagnosed with breast cancer, but have at least a 3% risk of developing the disease within 5 years.
“The USPSTF recommends against the routine use of risk-reducing medications, such as tamoxifen, raloxifene, or aromatase inhibitors, in women who are not at increased risk for breast cancer… This recommendation applies to asymptomatic women 35 years and older, including women with previous benign breast lesions on biopsy such as atypical ductal hyperplasia or lobular hyperplasia, and LCIS. This recommendation does not apply to women who have a current or previous diagnosis of breast cancer or ductal carcinoma in situ.” In this group of patients, the side effects associated with taking these medications would likely outweigh the prevention benefit.
The USPSTF based its recommendations on the evidence of the benefits and potential side effects of taking any of these risk-reducing medications, and an assessment of the balance. The USPSTF does not consider the cost of care in any of their assessments.
The USPSTF also states that it recognizes that clinical decisions involve more considerations than evidence alone. We strongly encourage a shared decision-making approach between patients and their healthcare team to determine what’s best for you. If you would like to learn if you could benefit from taking a risk-reducing medication and whether taking one is right for you, please discuss the full range of options, risks and benefits with your physician.
You can read the full USPSTF statement here.
FDA Calls For Men To Be Included In Breast Cancer Clinical Trials
Although breast cancer in men is not often publicized in the media, it is very important to know that men are at risk of the disease too. It is estimated that about 2,600 men will be diagnosed and 500 will die of breast cancer each year. However, because men contribute less than 1% of all cases, they are typically not included in clinical trials for treatment.
Since men have not historically been included in clinical trials, it is very possible they are not receiving optimal care. The drugs and treatment options they are prescribed have only been tested and approved for women.
The Food and Drug Administration (FDA) is calling for this to change. In their recent draft guidance, the FDA recommends the inclusion of men in breast cancer clinical trials to help improve the clinical management of the disease in male breast cancer. The FDA also states that if researchers propose to exclude male patients from a breast cancer trial, “scientific rationale should be included.” Furthermore, the FDA will not consider “low expected accrual rates of male patients with breast cancer” as sufficient rationale for excluding male patients moving forward.
The move seems to be working already… Following the FDA’s announcement, some breast cancer drug studies have extended eligible enrollment criteria to men.
USPSTF Expands BRCA Gene Testing But Not Far Enough
The US Preventive Services Task Force (USPSTF) issued a new recommendation statement calling for more patients to receive genetic counseling and genetic testing for BRCA1 and BRCA2 gene mutations.
Often referred to as the “breast cancer gene,” mutations in BRCA1 and BRCA2 genes have been shown to increase the likelihood of an individual developing breast, ovarian, fallopian tube and peritoneal cancer.
To better predict and manage the risk of developing hereditary cancers, the USPSTF is now recommending more patients, including breast, ovarian, fallopian tube and peritoneal cancer survivors, undergo genetic counseling and genetic testing. Previously, the USPSTF only recommended those who had a family history BRCA-related cancers be tested.
The USPSTF statement, published in the JAMA Network, recommends that “primary care clinicians assess women with a personal or family history of breast, ovarian, tubal, or peritoneal cancer or who have an ancestry associated with BRCA1/2 gene mutations with an appropriate brief familial risk assessment tool. Women with a positive result on the risk assessment tool should receive genetic counseling and, if indicated after counseling, genetic testing… The USPSTF recommends against routine risk assessment, genetic counseling, or genetic testing for women whose personal or family history or ancestry is not associated with potentially harmful BRCA1/2 gene mutations.”
Why are these tests important? Expanding testing will potentially help survivors determine which treatments would be best based on their current diagnosis, as well as alert them to treatments that could significantly lower their future cancer risk, such as risk-reducing mastectomy and hysterectomy. It could also be very important and possibly life-saving for some of the survivors’ family members.
While the updated recommendations are very good news, in several ways they do not go far enough: newly diagnosed patients with breast or ovarian cancer, patients with advanced cancers, and men were not included. Men who carry a BRCA gene mutation are at increased risk for breast, pancreatic and prostate cancers, and should also have counseling and testing. As in women, these mutations can be passed on to their children and beyond.
Anyone who is concerned about their future cancer risk and doesn’t fall into these guidelines should consider proactively seeking a consultation with a genetic counselor. You can find a certified genetic counselor near you via the National Society of Genetic Counselors.
New Blood Test could predict Breast Cancer Recurrence and even prevent Surgery
While there is still much work to be done, detection and treatment of early stage breast cancer have improved tremendously over the last few years. Chemotherapy regimens have become increasingly effective, boosted by the emergence of immunotherapy, and often lead to a ‘pathological complete response’ when given before surgery (known as ‘neoadjuvant therapy’). A pathologic complete response means there is no identifiable tumor in the tissue that is resected by the lumpectomy or mastectomy.
However, there is currently no way to truly know which patients treated for early stage breast cancer enter remission and which ones actually have residual cancer cells in their system (in the absence of obvious metastatic disease).
A recent study published in Science Translational Medicine reported very encouraging news on a test called Targeted Digital Sequencing (or ‘TARDIS’) that could potentially help predict which patients are at high risk of breast cancer recurrence. TARDIS identifies DNA released by breast cancer cells into the bloodstream but is up to 100 times more sensitive than other liquid-biopsy tests currently available or in development.
This could be a huge step forward since TARDIS will potentially help doctors identify patients with early stage breast cancer who may still have residual cancer cells in their bodies after treatment that aren’t otherwise detectable by standard scans and follow-up monitoring.
It is also possible that the test could help remove the need for surgery altogether in women who experience a pathological complete response with neoadjuvant chemotherapy and are shown to have no residual breast cancer DNA in their bodies.
Very exciting news indeed! Read the full article here.
Allergan Recalls its Natrelle BIOCELL Textured Breast Implants and Tissue Expanders due to risk of BIA-ALCL
On July 24th, Allergan announced it was recalling its BIOCELL textured expanders and implants from the global market in response to a request from the FDA. The FDA made the request that Allergan recall all its BIOCELL textured breast implants and tissue expanders based on newly submitted Medical Device Reports reporting worldwide cases of Breast Implant Associated Anaplastic Large-Cell Lymphoma (BIA-ALCL), and related deaths associated with these devices.
BIA-ALCL is a rare cancer of the immune system that develops in the scar tissue surrounding breast implants. Common symptoms include swelling, significant fluid accumulation around the implant, pain and/or lumps in the breast. When caught early, the cancer is curable by removing the breast implant and the surrounding scar tissue (known as a capsulectomy). Patients with advanced disease need more treatment in the form of chemotherapy and radiation.
To date, 573 cases of BIA-ALCL and 33 deaths have been confirmed worldwide. Additional cases have been reported but have not been confirmed. Of the 573 confirmed cases, 481 have been attributed directly to Allergan’s Biocell textured implants leading to a global recall of the devices.
The US recall comes after an official FDA hearing was held in March to evaluate textured breast implant safety. The FDA first reported the possible link between breast implants and anaplastic large cell lymphoma in 2011. All confirmed cases have been attributed to textured implants. While there have also been cases in women who had smooth implants, due to the lack of a comprehensive medical history and incomplete implant information in these patients, there have been no confirmed cases attributed solely to smooth implants.
Although the Allergan textured implants have been recalled, the FDA is not recommending preventative removal of these textured implants in women who don’t have any symptoms due to the very low risk of developing BIA-ALCL. The FDA does recommend patients be educated on the potential risks associated with textured implants as well as the signs and symptoms of any complications. Patients are encouraged to contact their plastic surgeons with any questions or concerns whatsoever about their implants.
Read the full FDA patient safety communication here.
Will this detect breast cancer 5 yrs earlier?
MIT’s Computer Science and Artificial Intelligence Lab has developed a new tool that uses artificial intelligence (AI) and deep learning to predict the development of breast cancer up to five years earlier than current detection techniques.
Unlike other AI-based tools that use data mostly from acquired from white patients and may therefore be algorithmically biased, MIT’s model works equally well for both white and black patient populations. This is particularly important as black women are 42% more likely to die from breast cancer than white women – a possible contributing factor could be that current detection techniques don’t work as well in black women.

To create the tool, data was obtained from over 90,000 mammograms and outcomes from over 60,000 patients treated at the Massachusetts General Hospital. A form of machine learning known as “deep learning” was then used to identify patterns in the images and data that are too subtle to be routinely recognized by human physicians. The results have so far have been more accurate than current diagnostic approaches, presumably because the model is not based on existing knowledge and assumptions about patient risk factors.
MIT’s project is intended to help doctors compile the best personalized screening program for their patients and hopefully eliminate the heartbreaking outcome of a late breast cancer diagnosis. We eagerly await to hear if and when this could become widely available.
New surgical guideline leads to lower re-operation rates after lumpectomy surgery
Re-operation rates following breast conservation (lumpectomy and radiation) for early invasive breast cancer have been highly variable historically, mainly because of uncertainty and variability in what surgeons deemed safe.
When a tumor is removed, the specimen is painted with a special ink before it is evaluated by a pathologist. This ink allows the pathologist to clearly see the outer edges, or ‘margins’, of the tissue under the microscope.
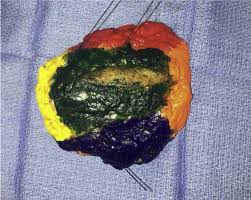
In 2014, the Society of Surgical Oncology and American Society for Radiation Oncology released new guidelines stating that as long as the tumor has no ink on it, the margin is clear. A clear margin means there are no cancer cells at the edge of the surgical specimen and tells the surgeon that all the cancer was removed.
Prior to the new guidelines, some surgeons wanted at least 2mm of normal breast tissue around the cancer. Others accepted less. Even though wider clear margins don’t reduce the risk of cancer recurrence, because of this lack of consensus, 25%-30% of patients having a lumpectomy required more surgery to ensure a larger clear margin.
A recent study in the American Journal of Surgery shows that the new guidelines have led to a decrease in re-operation rates. This offers peace of mind that you likely won’t need another “re-excision” surgery after a lumpectomy if the margins are clear, irrespective of how small the clear margins are, as long as there is “no ink”. Additionally, surgeons now have an evidence-based standard to follow. Very reassuring news indeed!




